Products
Filler Background
For many years, traditionally cross-linked hyaluronic acid of non-animal origin has been a proven substance in dermal fillers and has been shown to be very safe and effective.
Thanks the innovative spherification technology, a result of many years of Swedish research, we can now introduce a hyaluronic acidbased dermal filler with smooth, spherical particles instead of the traditional structure with angular rhomboids.
The name of the dermal filler is SweDermis, an advanced product in which the smooth spherical particles contribute to an even smoother result – and greater patient satisfaction.







The New Regulatory Standard for Dermal Fillers
Moving Beyond CE-MDD to MDR
In modern aesthetic medicine, the safety of dermal fillers is no longer simply a competitive advantage. It is a scientific, legal, and regulatory requirement. With the implementation of the European Medical Device Regulation (MDR), the standards governing the development, manufacture, and approval of medical devices, including dermal fillers, have become significantly more demanding across Europe.
This regulatory shift has increased the responsibilities of manufacturers while raising the level of safety, traceability, and transparency expected throughout the aesthetic medicine sector.
1. Transition from MDD to MDR
From directive to binding regulation Previously, many medical devices entered the European market under the Medical Device Directive (MDD). That framework focused mainly on the initial technical documentation required at the time of market entry, and it allowed manufacturers to rely on the concept of equivalence. In other words, approval could sometimes be supported by demonstrating similarity to existing products.
With the introduction of MDR, that model changed fundamentally. MDR is a binding regulation that imposes detailed oversight across the entire lifecycle of a medical device.
It covers every stage of development and use, including:
• Product design and development
• Biocompatibility assessment
• Detailed analysis of chemical composition and material structure
• Evaluation of manufacturing processes
• Product-specific clinical evidence
• Post-market surveillance and ongoing safety monitoring
Because dermal fillers are implantable substances that remain in the body, they are subject to particularly strict scrutiny under MDR. This results in a substantially higher level of long-term safety evaluation.
As a consequence, many legacy products that cannot meet the new requirements are gradually being removed from the European market.
2. Why MDR Certification
MattersAdvantages for distributors, physicians, and clinical users For pharmaceutical distribution companies, commercial partners, physicians, and professionals who use dermal fillers in clinical practice, MDR certification represents a meaningful level of assurance and reliability.
Its key advantages include:
• Proven clinical integrity. MDR requires direct clinical evidence for the specific product itself. Safety and performance must therefore be supported by dedicated data for that exact filler, rather than by reference to similar products.
• Full product traceability. Through the Unique Device Identification (UDI) system, every syringe can be traced throughout the supply chain. This supports authenticity, transparency, and rapid action if any safety issue arises.
• Continuous safety monitoring. Under MDR, safety oversight does not end after market approval. Manufacturers are required to maintain post-market surveillance and submit Periodic Safety Update Reports (PSUR) to monitor and manage potential adverse events over time.
3. Demonstrating Technical Superiority
Scientific and manufacturing excellence Achieving CE-MDR certification is a demanding and highly complex process. Only companies with strong scientific infrastructure, advanced manufacturing systems, and sustained regulatory capability are able to complete this pathway successfully.
During this process, a dermal filler is subjected to extensive technical review,
including:
• Detailed evaluation of chemical composition and material architecture
• Comprehensive review of manufacturing processes and quality control systems
• Assessment of raw material purity and stability
• Analysis of clinical data and clinical outcomes
• Ongoing safety and performance testing
In advanced fillers based on hyaluronic acid, the purity of the raw material and the precision of molecular modification are critical. High-purity bio-fermented hyaluronic acid, combined with a controlled formulation approach and minimal unnecessary additives, supports better biocompatibility and helps reduce the risk of unwanted inflammatory responses.
This level of scientific control and manufacturing precision contributes to stronger physician confidence, greater trust among clinical users, improved market positioning, and more stable product continuity.
4. Regulatory Validation in Sweden
In addition to MDR compliance, our products are supported by regulatory recognition associated with the Swedish Medical Products Agency (Läkemedelsverket).
The Swedish Medical Products Agency is widely regarded as one of the most respected and demanding regulatory authorities in Europe in the fields of pharmaceuticals, medical devices, and healthcare products.
Recognition connected to this authority reflects adherence to very high standards of safety, manufacturing quality, and scientific review. For healthcare and aesthetic products, this level of oversight is internationally associated with rigorous regulatory expectations.
5. Swedermis – Swedish Precision in Dermal Fillers
Built on these strict scientific and regulatory foundations, Swedermis dermal fillers, developed in Sweden, are designed to meet the expectations of physicians, aesthetic clinics, distributors, and professional users who require reliable, high-quality filler solutions.
Key characteristics include:
• High-purity hyaluronic acid
• A controlled and refined formulation profile
• Advanced gel manufacturing technology
• Strong tissue integration and structural stability
• Predictable and reliable injection performance
By combining scientific research, precision manufacturing, and strict regulatory compliance, Swedermis has been developed as a dependable dermal filler solution for modern aesthetic medicine.




SweDermis is available in four versions:
The SweDermis range includes four formulations, each designed for specific facial indications and treatment needs.
SweDermis Line
SweDermis Lips
SweDermis Fill
SweDermis Lift
Area of use
SweDermis Line
- Frown lines
- Perioral lines
- Labial commissure
- Cupid’s bow
- Fine facial lines
- Superficial wrinkles
- Lip contour definition
SweDermis Fill
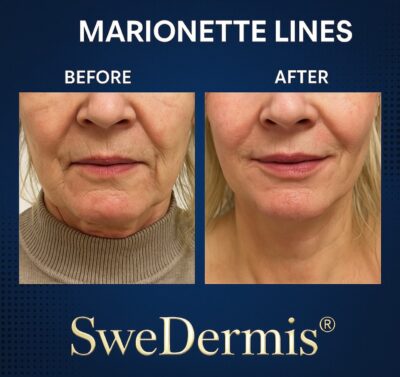
- Nasolabial folds
- Marionette lines
- Cupid’s bow enhancement
- Mid-face volume restoration
- Moderate facial contouring
- Facial wrinkle correction
SweDermis Lips
- Lips
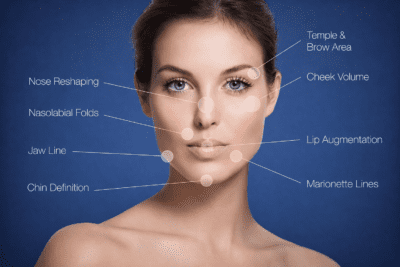
SweDermis Lift
- Facial volumizing
- Cheek enhancement
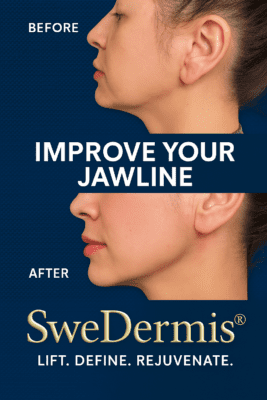
- Jawline definition
- Chin shaping
- Deep facial contouring
- Structural facial support
Application
SweDermis Line should be injected intra- dermally through a needle into mid dermis. SweDermis Lips should be injected intra- dermally through a needle into the deep layer of the dermis and/or the surface lay- er of the subcutis. The depth of injection with SweDermis Lift may vary between injections into the sub- cutaneous tissue to supraperiosteal ad- ministration depending on the treatment site.
Product description
SweDermis products are composed of cross- linked hyaluronic acid of non-animal origin and are supplied in a graded, disposable, 1.0 ml glass syringe. Two disposable sterile needles are provided with each syringe. The products are for single use only.

Digital Brochure

Digital leaflet

